Cdrh Organization Chart
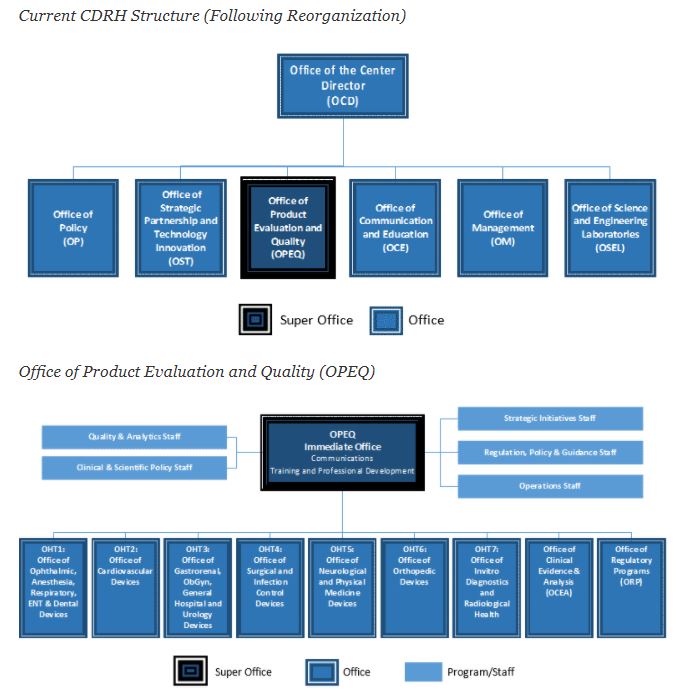
Cdrh Organization Chart - Issued may 2, 2002 the fda's center for devices and radiological health (cdrh) works to ensure public safety by certifying the safety and effectiveness of medical and radiologic devices in. Web free essays, homework help, flashcards, research papers, book reports, term papers, history, science, politics The following header reflects the organizational hierarchy. The program contributes to the center’s mission of protecting and promoting public health through the development and recognition of voluntary consensus standards that serve to establish safe and effective. Web keith began by sharing the current cdrh organizational chart, which was finalized in october 2019. Cdrh management directory by organization. Web per the cdrh release, its primary functions include an external focus on issues such as “medical device cybersecurity, digital health, standards, and patient science,” and coordinating the center’s readiness for crisis events such as pandemic outbreak or supply chain shortages. At the top of this structure, you'll find the office of the center director. Web cdrh office of communication and education organization chart food and drug administration office of medical products and tobacco center for devices and radiological health office of communication and education. Web • cdrh reorganization includes adopting a tplc model and other efforts to streamline and improve efficiency and to support professional growth • implementation timeline: 3 old cdrh structure super office office division branch team program/staff office of the center director The cdrh standards program was established as a result of the food and drug administration modernization act (fdama) of 1997. Web the cdrh organizational chart is updated quarterly. Web previous cdrh organization model. Although many of the senior staff within these new offices will. Web keith began by sharing the current cdrh organizational chart, which was finalized in october 2019. Submitting reports and requirements for maintaining records for. The cdrh standards program was established as a result of the food and drug administration modernization act (fdama) of 1997. Cdrh mailing addresses and office phone numbers; 3 old cdrh structure super office office division branch. Web though there are additional changes observed in the organizational chart, those discussed above will likely be of most importance to u.s. Web content current as of: Department of health and human. Submitting reports and requirements for maintaining records for. Reorganization of the center for devices and radiological health. Web keith began by sharing the current cdrh organizational chart, which was finalized in october 2019. She pointed out two new offices that are the result of programs and functions that were spun out of the office of the center director―the office of policy, and the office of strategic partnerships and technology innovation (see figure 1 ). 3 old cdrh. Web the current cdrh structure, following reorganization, follows: Cdrh mailing addresses and office phone numbers; Submitting reports and requirements for maintaining records for. 3 old cdrh structure super office office division branch team program/staff office of the center director This list enables you to view charts of performance data and progress on important projects and programs. Cdrh management directory by organization; Web keith began by sharing the current cdrh organizational chart, which was finalized in october 2019. Web per the cdrh release, its primary functions include an external focus on issues such as “medical device cybersecurity, digital health, standards, and patient science,” and coordinating the center’s readiness for crisis events such as pandemic outbreak or supply. Web keith began by sharing the current cdrh organizational chart, which was finalized in october 2019. Web the cdrh organizational chart is updated quarterly. The cdrh standards program was established as a result of the food and drug administration modernization act (fdama) of 1997. She pointed out two new offices that are the result of programs and functions that were. Web the current cdrh structure, following reorganization, follows: Web the list below provides information on fda's performance measures and projects as aligned to fda strategic priorities and program offices. Department of health and human. At the top of this structure, you'll find the office of the center director. Cdrh mailing addresses and office phone numbers; Reorganization of the center for devices and radiological health. Organization chart for the fda's center for devices and radiological health, including leadership roles. Department of health and human. Submitting reports and requirements for maintaining records for. Cdrh management directory by organization; Web • cdrh reorganization includes adopting a tplc model and other efforts to streamline and improve efficiency and to support professional growth • implementation timeline: Reorganization of the center for devices and radiological health. The center for devices and radiological health (cdrh) is the branch of the united states food and drug administration (fda) responsible for the premarket approval of. Web cdrh office of communication and education organization chart food and drug administration office of medical products and tobacco center for devices and radiological health office of communication and education. The program contributes to the center’s mission of protecting and promoting public health through the development and recognition of voluntary consensus standards that serve to establish safe and effective. Medical device and diagnostic manufacturers. Web keith began by sharing the current cdrh organizational chart, which was finalized in october 2019. Web • cdrh reorganization includes adopting a tplc model and other efforts to streamline and improve efficiency and to support professional growth • implementation timeline: Center for devices and radiological health. The following header reflects the organizational hierarchy. Organization chart for the fda's center for devices and radiological health, including leadership roles. She pointed out two new offices that are the result of programs and functions that were spun out of the office of the center director―the office of policy, and the office of strategic partnerships and technology innovation (see figure 1 ). Web the current cdrh structure, following reorganization, follows: Web though there are additional changes observed in the organizational chart, those discussed above will likely be of most importance to u.s. Web content current as of: At the top of this structure, you'll find the office of the center director. Web previous cdrh organization model. The cdrh standards program was established as a result of the food and drug administration modernization act (fdama) of 1997. The following bullets capture the cdrh management directory, by organization: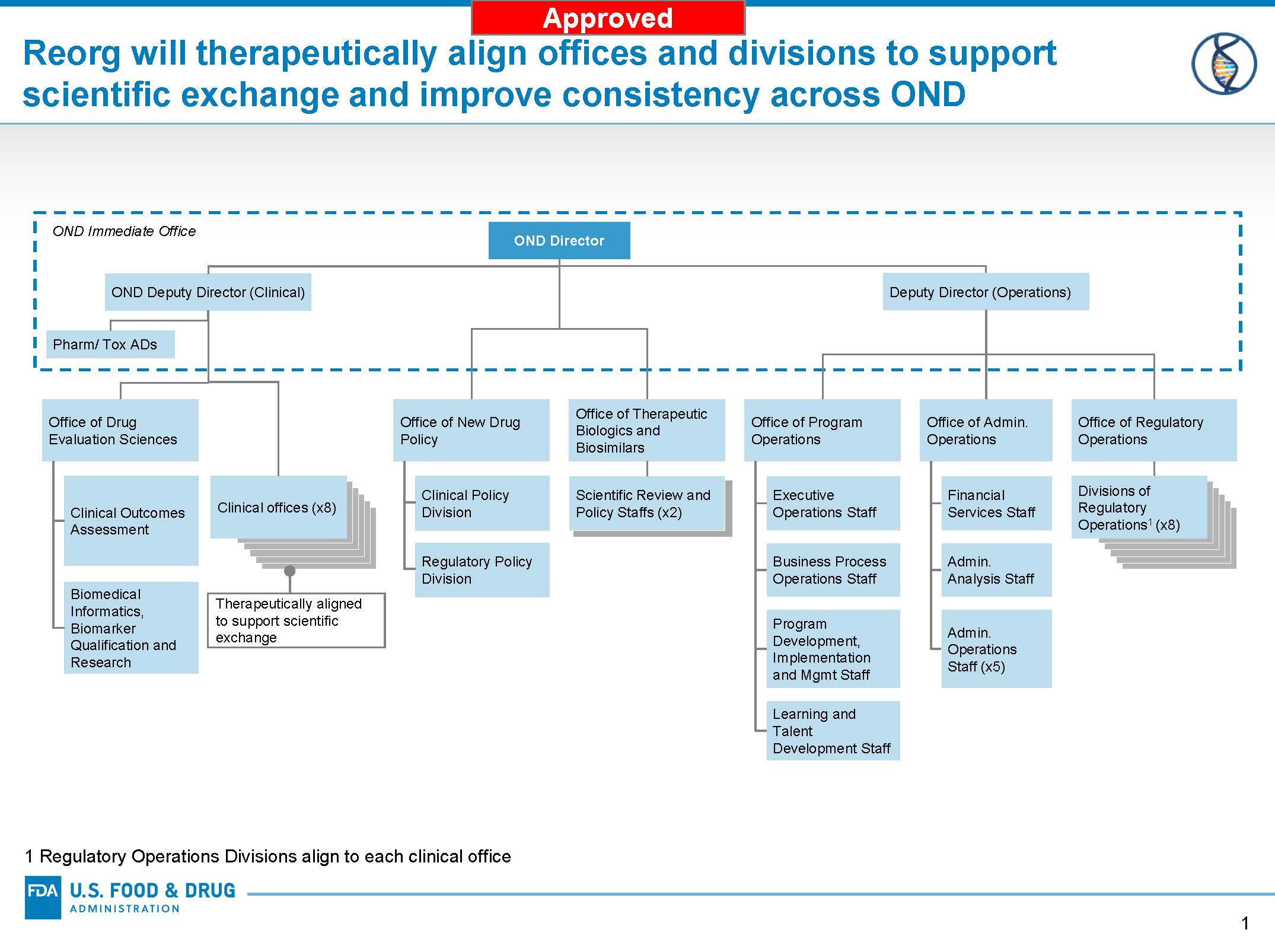
Of The Office Of New Drugs With Corresponding Changes To

It’s Now Official The New CDRH Organizational Structure and How It May
Center For Device and Radiological Health Dawnbreaker MRR

Organizational Structure of the FDA. Download Scientific Diagram

Download Template Desain Kemasan Produk Cdrh Organizational Chart
Fillable Org Chart Learn Diagram vrogue.co

PreApplication Information Webinar for PAR21183, "Developing Digital

FDA CDRH Organizational Structure & Overview
Operational Organizational Chart
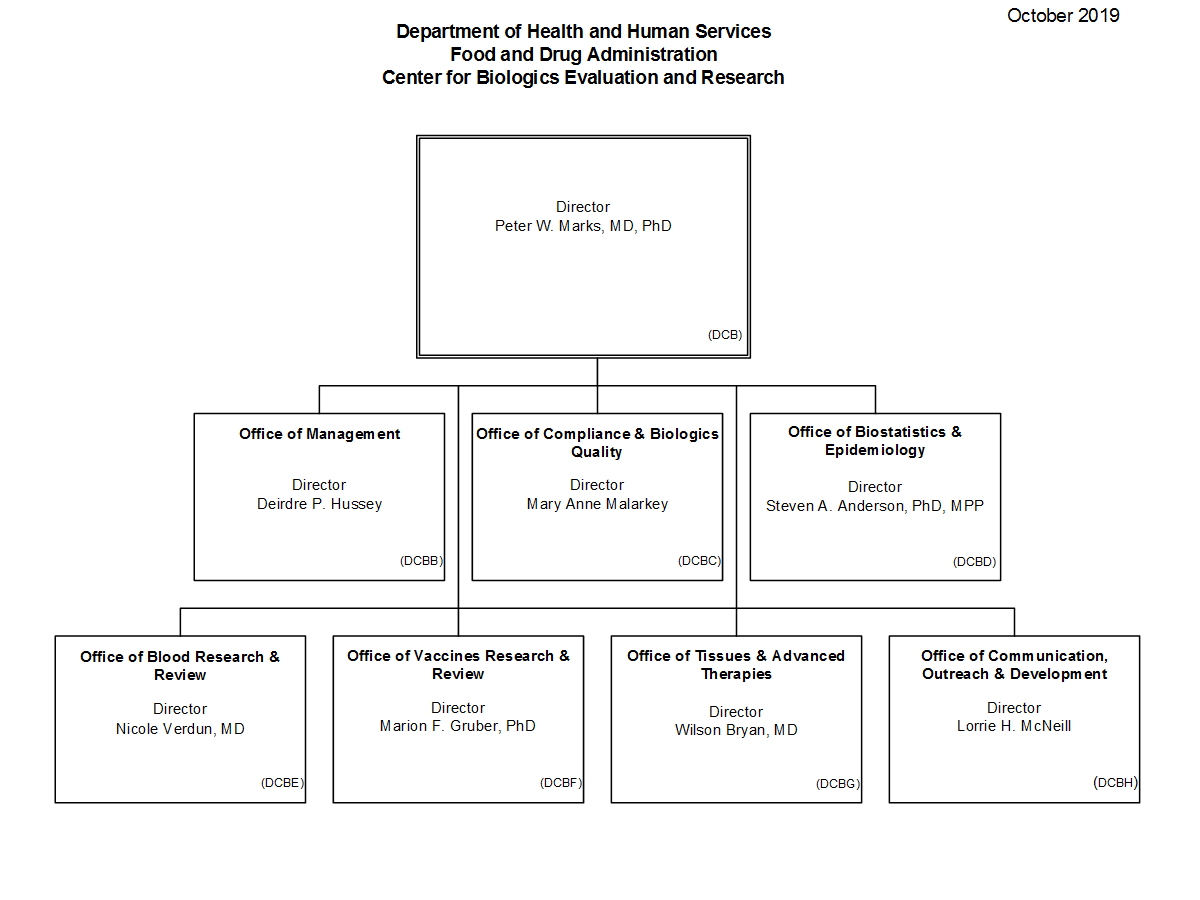
Center for Biologics Evaluation and Research Organization Chart FDA
Cdrh Management Directory By Organization;
For Specific Phone Numbers And Email Addresses For Each.
Although Many Of The Senior Staff Within These New Offices Will Remain The Same, They Will Now Have New Titles.
Web Per The Cdrh Release, Its Primary Functions Include An External Focus On Issues Such As “Medical Device Cybersecurity, Digital Health, Standards, And Patient Science,” And Coordinating The Center’s Readiness For Crisis Events Such As Pandemic Outbreak Or Supply Chain Shortages.
Related Post: